The common fruit fly’s pruning protein, which controls the metamorphosis of the animal, may one day have implications in reversing brain injuries, according to Duke University researchers. The protein directs both early development and regrowth of dendrites that relay information from neuron to neuron. The university points out in a recent news release that if a neuron is injured or malformed, they frequently don’t have the proper dendrites needed to be functional.
Chay Kuo, MD, PhD, the George W. Brumley, assistant professor of cell biology, neurobiology and pediatrics, states that a major issue within the nervous system is that it does not regenerate well following injury, “Neurons don’t multiply, so when they’re injured, there’s a loss of function. We’d like to know how to get it back.”
The branching out of dendrites from the body of the neuron are a necessary part of the fruit fly’s Drosophilla’s life cycle. Kuo says that until now, researchers have not understood how drosophila neurons are able to create two separate dendrite branching systems that serve different kinds of sensory environments. In the larval state, the fly’s nervous system is tuned into the needs of the smooth-skinned worm such as finding food, locomotion, and avoiding attack. As an adult that has developed bristle-covered skin, the nervous system is now wired for flying, finding mates, and laying eggs.
Kuo states that the key to this transition is the insect’s metamorphosis from larvae to adult. During the transition, Drosophilla lose the neurons that are not necessary to adult life. Remaining sensory neurons then sever their dendrites and grow a completely different set.
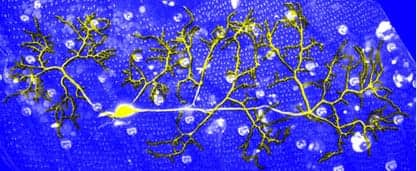
To determine the genetic mechanism that makes possible, Kuo and his team first sought to pinpoint how drosophila sensory neurons accomplish this change. The team reportedly tagged abdominal sensory neurons with green fluorescent protein (GFP) and followed them through metamorphosis to confirm if their dendrite branching had changed. The results indicate that the dendrite design and architecture was different in the adult stage.
According to test results obtained by Gray Lyons, graduate student, Cysteine proteinase-1 (Cp1) is responsible for regulating the regeneration of neuron dendrites and innervating the adult sensory field. Without Cp1, the team says, drosophila sensory dendrites cannot regenerate after pruning.
Resarchers explain that the mammalian version of Cp1 is lysosomal protein capthesin-L (Ctsl). During the cell cycle, Ctsl can target the protein Cut-like 1 (Cux1) that plays a role in gene expression. Ctsl pursues Cux1 inside the nucleus and cuts it, creating a smaller protein with different transcriptional properties than the original one, the researchers say.
The release states that Kuo’s team tagged Cut during Drosophila metamorphosis and observed the protein’s binding pattern within the nucleus. Prior to dendrite pruning, Cut binds in large blobs, the team says. Following pruning, Cut binding is diffused, giving it an opportunity, Kuo explains, to bind to different genes during the two dendrite growth phases.
The team then translated this finding back to Cp1, the release says, discovering that it goes into the neuron nucleus to cleave Cut, making a new transcription factor required for dendrite regeneration after developmental pruning.
The findings may impact the understanding and treatment of brain injuries, if researchers can one day help neurons modify their original developmental program and regrow new dendrites. “If we can influence this environment control that changes the development program, it’s possible that we could get neurons to integrate and function better after injury,” Kuo says.
Photo Credit: Chay Kuo Lab, Duke University
Source: Duke University